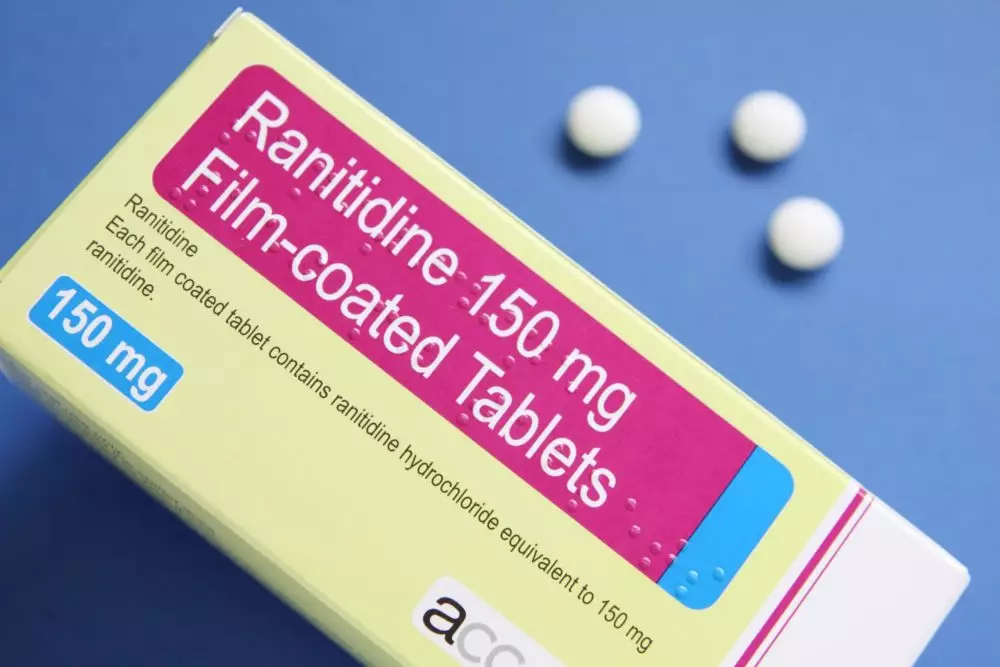
Ranitidine recall 2020: FDA asks for immediate withdrawal of all ranitidine drugs (such as Zantac) - oregonlive.com

Information Update - Additional ranitidine products recalled, including Zantac; request to stop distribution remains in place while Health Canada continues to assess NDMA